From Friday, April 19th (11:00 PM CDT) through Saturday, April 20th (2:00 PM CDT), 2024, ni.com will undergo system upgrades that may result in temporary service interruption.
We appreciate your patience as we improve our online experience.
From Friday, April 19th (11:00 PM CDT) through Saturday, April 20th (2:00 PM CDT), 2024, ni.com will undergo system upgrades that may result in temporary service interruption.
We appreciate your patience as we improve our online experience.
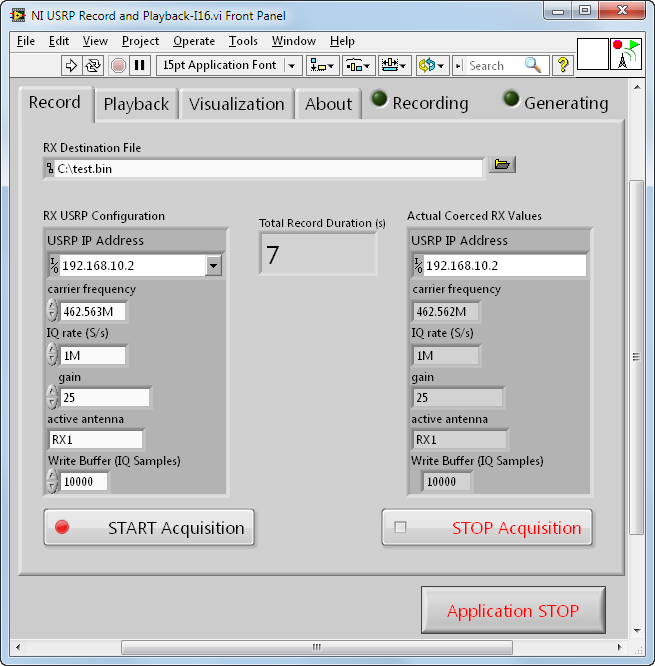
Choose your file path, IP address, Carrier Frequency, IQ Rate, Gain, and Antenna (on the left) and you're ready to Record.
Be sure to observe the Actual Coerced values so that you know exactly what parameters were used for the actual recording.
See the Record and Playback demo video and tutorial
Download the NI USRP 1.1 driver
Could you please attach the niUSRP Fetch Rx Data (I16).vi and niUSRP Write Tx Data (I16).vi ?
Thanks
Those VIs are available with the NI-USRP 1.1 release, which we'll be posting online in the next few days.
Hi,
I have used the NI USRP Record and Playback - I16.vi to record the GPS signal from a PXIe-8106 (include PXIe-5442 100 MS/s AWG OSP, PXI-5610 RF-Upconverter, Averna Dynamic Range Extender). The GPS Signal is generated by Averna NAVASIM (NAVigation Advanced SIMulator).
When I connect the RF Out Port from the Dynamic Range Extender to a Signal Analyser the following signal can be seen:
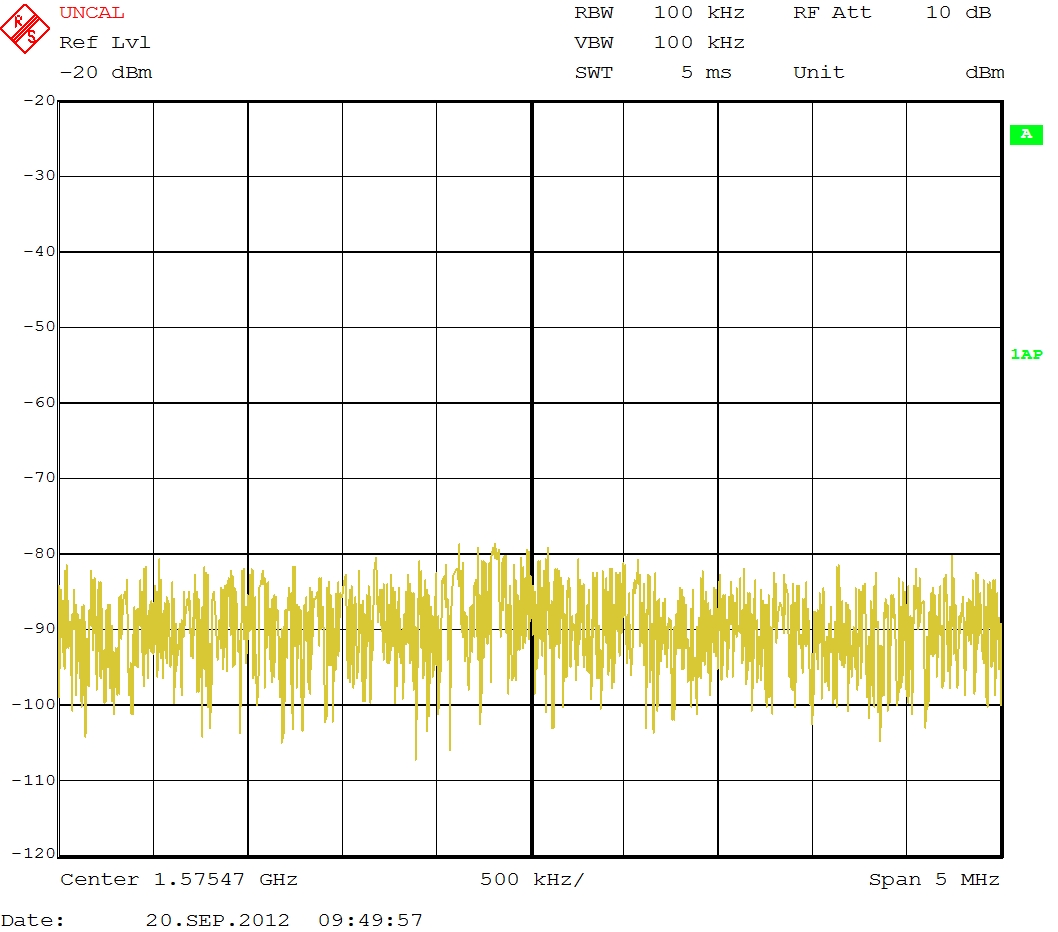
Averaged
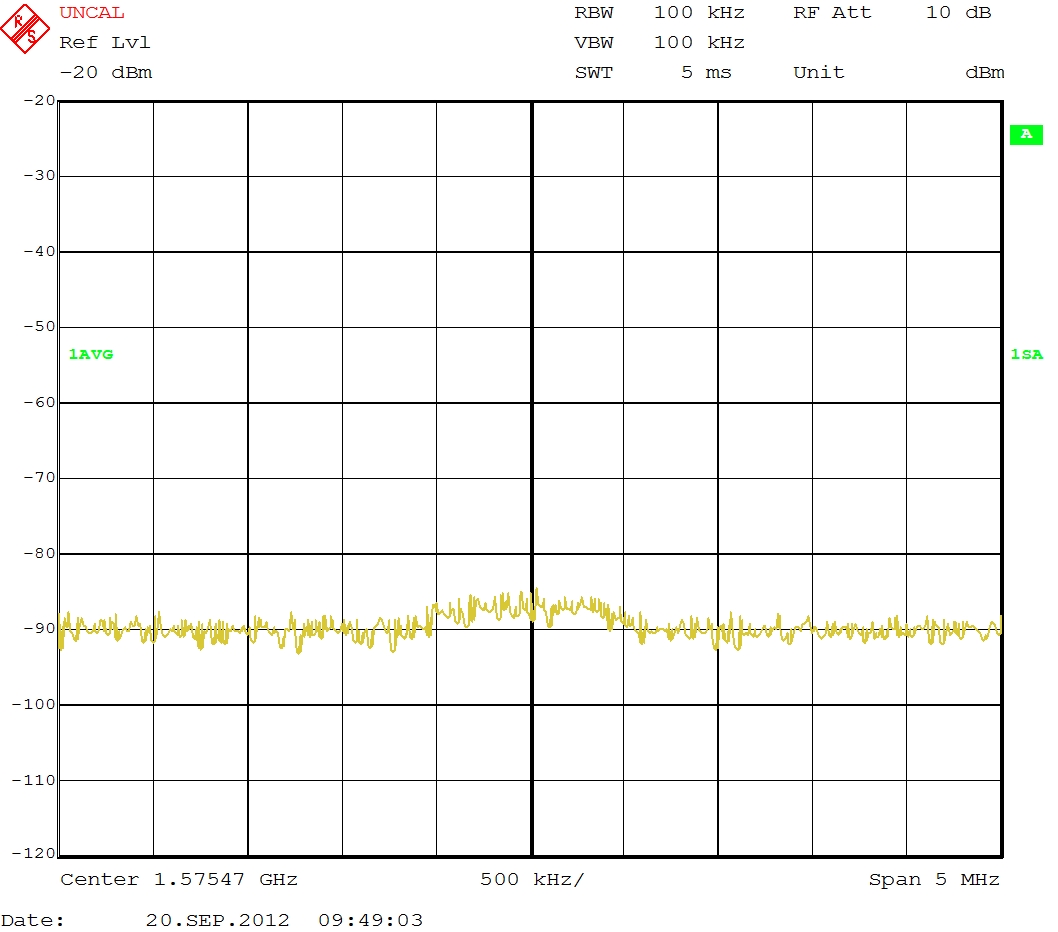
When I connect the RF Out Port from the PXI-5610 RF-Upconverter to a Signal Analyser the following signal can be seen:
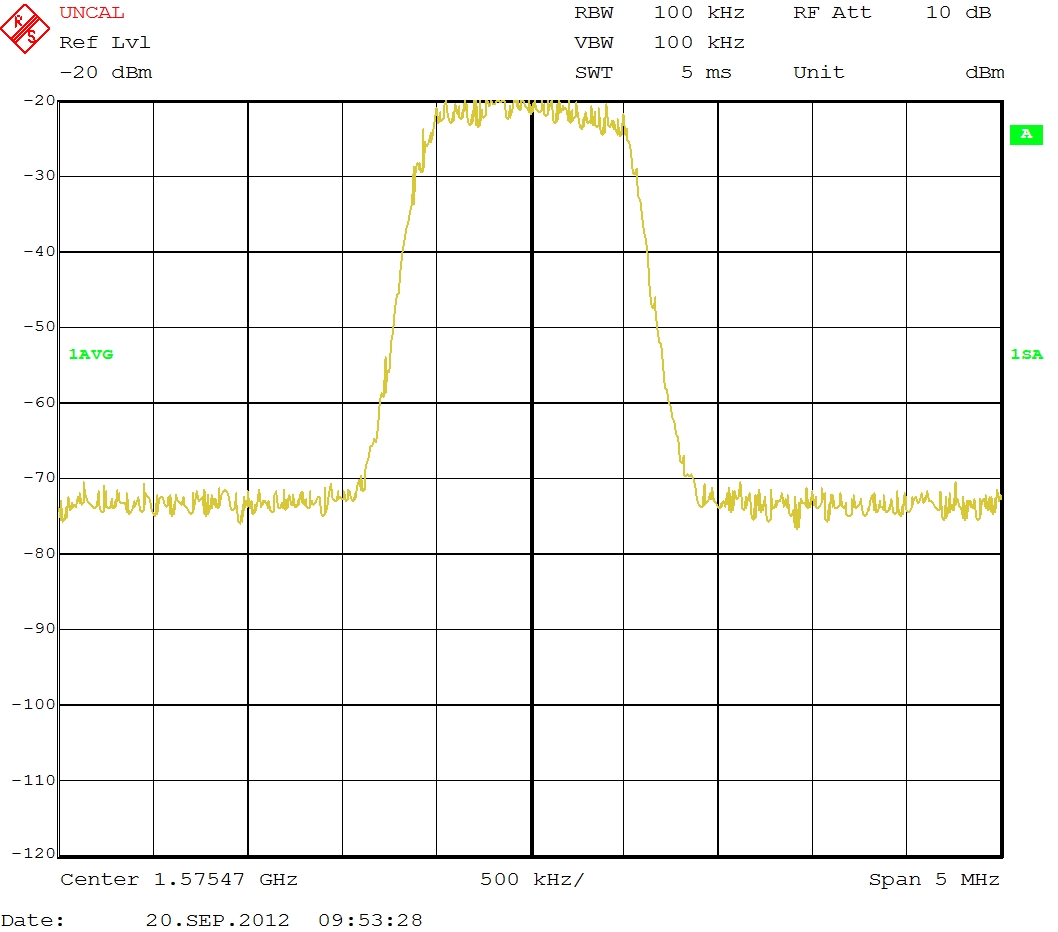
With the two 30dB attenuators from the the USRP package in line the signal is like this:
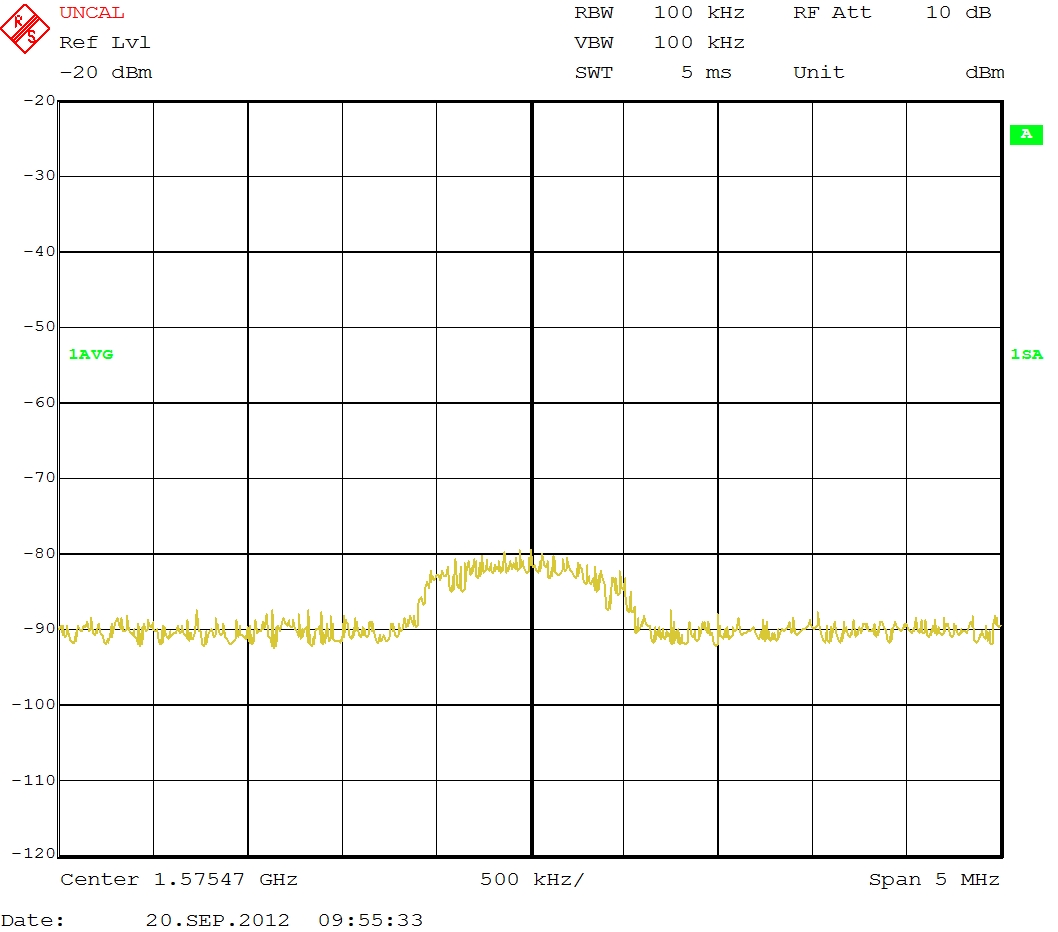
I have recorded GPS signals from these three configurations, but when I playback all the recorded signal with the USRP the signal looks like this on the signal analyser:

Does anybody have a idea?
In the demo video a ublox was used to record the gps signal while driving. How is it possible to record with u-center in a binary data format which is useable inside LabView for playing back the gps signal? Ublox uses the *.ubx data format, is there a converter?
Hello,
Since we have an USRP in our office, we tried to reproduce the GPS record and playback with a Ublox evalution Kit. However, the RF input of this one has a 3,3V dc to supply a GPS antenna. Did you just use attenuators and a exterior reference for the clock or did you add a DC-Block between the USRP and the Ublocx? Is the USRP protected against this kind of thing ?
The output power of the USRP is very strong (and does not have protection). We used three (3), 30dB attenuators and transmitted with a power level of 0 in the software.
Erik
the USRP was used for both record and playback. The uBlox was simply used for verifying that playback was accurate enough to allow a GPS (the uBlox) to lock.
Hi,
I tried to reproduce the record and playback with the VIs you gave in the top of this page. Unfortunately, I am unable to lock a GPS signal. I recorded the signal from my office (GPS antenna outside) for 60 seconds at least.
Here is the configuration for the record:
- GPS antenna,
- BIAS Tee from mini circuit: ZFBT-4R2G-FT+
- Amplifier from mini-circuit: ZX60-33LN-S+ (+16 dB)
- An external clock reference
Here is the configuration for the generation:
- Ublox (various tests with 90 to 60 dB of attenuation)
- An external clock reference
Whatever the attenuation, no satellite can be acquired nor tracked.
The configuration for the computer is the following :
Tablet PC with Intel(R) Core(TM)2 Duo CPU U7700 @1,33GHz, 1,75 Go RAM and ethernet card : Broadcom NetXtreme 57xx Gigabit.
What did we do wrong ? Did we forget something? We tried all the tests with the original Vis. Something is wrong with them? Any idea?
Hi Cheetor,
I think 60 secods are not enough. My recordings were at least 15 min. You should also use a better notebook. In my case I have used a Core2Duo 1,8 GHz, 4GB RAM and I had several problems with playback (underflow erroers). These problems emerged from simultaneous activities like web browsing.
You may also need to add a property node to the RX and TX config in order to select the external clock as the frequency reference.
Hi,
Thanks for your help.
I tried with the property node. Unfortunately, it is still not working. Any idea?
What did you use for the record and playback configuration ? I tried the record with IQ rates of 2M, 2,5M and with a gain between 0 and 25 dB.
Did you configure something special in the u-blox software ?
I've used 3MS/s and 5MS/s. I used the 3rd tab to see the incoming signal and it was a nice wide curve in the center of the screen with a width of about 2 MHz. I also used the unlocked with the antenna to test the antenna. Then I used the bias tee with a 30db attenuator to test that section of connections again with the GPS. Then I attached to RX1 (verify antenna port is correct) and recorded at15-25 dB internal amplification. Externally I used the bias T and an additional 30db amplifier both from mini circuits. I did use a GPS filter before going to the but I did not appear to be critical. I also used a simple USB power meter and tx'ed a single tone to get the 0 and 30dB output power of my USRP. At GPS frequencies it was about -15dBm to 15dBm respectively on a USRP-2920. Also it isinteresting that if you set the record frequency wide enough you'll start to see other non-GPS GNSS signals.
Hi Erik,
Thanks for your help and your quick answers.
I've done the first step (see the incoming signal), with a bias T and an amplifier ( +16 dB for GPS carrier frequency). The record was set with the following values :
This is what we get with the Visualization tab :
I have test the amplifier, it works fine. Any idea ? Complementary test ?
I don't see the GPS signal I expect. It should look like the oscilloscope screen shot in in YYY's comment above, second from the bottom (a hill rather than a spike). Maybe more amplification is needed, or a clearer view of the sky with your antenna would help.
Regards,
Erik
Can we change the recording quantization level to 8 bit per sample intead of 16 bit?
Thanks.
Hi,
I came back to you with an other question. From time to time, when we play back the signal, the duration time appears to be different than the time of record. How is this possible ?
Thanks a lot for your answer.
I can't find where to download the playback .vi file ... where is it?
Whether I can use this function or vi to relay the information between source and destination? Thanks
8 to 16 bit can be changed with 4 steps for most efficient use. 1. use a property node to change the transport to 8 bit mode. 2. change the niUSRP Read and write to 8 bit. 3 change the queues used to store data to 8 bit mode by changing the datatypes to I8. And lastly use a property node to choose which 8 bits are most significant so you send those back. Check the help file for niUSRP for more info on 8 bit mode.
When I have used record and playback and the times have been different durations it was because the record and playback IQ rates were different.
The VI contains a tab control with several tabs. One tab is record. The next is playback. They look very similar.
The USRP can be used to make a relay. Your technique may differ a bit with an all wireless relay because you may not want to write data to disk.
when I set the IQ rate to 5M, and playback, here is a error:
Underflow: the Tx buffer was emptied before new data was provided. Consider reducing the IQ rate, increasing the Write rate, or increasing the number of samples per Write.
Three things to try:
1. Use the NI USRP 1.2 driver
2. Make sure your PC is running in "High Performance" mode in Windows power options
3. Use the vendor's Ethernet chipset driver rather than the generic windows default.
My laptop tips out between 8 and 10 MS/s. my desktop PC will do full 25 MS/s.
The processing in the loop and the speed of the hard drive make a big difference. Hard drives advertise peak write speeds but not sustainable. And the more fragmented the drive or the center of the spinning platter reads/writes slower.
Is it possible to record RF signals transmitted from radio station and playback later on? I actually modified visualization loop by including simple sound.vi. but I hear only noise when I playback? Any suggestions about this approach or isn't it possible to do that?
Thank you!
Hi,
Since I use an USRP as part of my course, I have a few questions about its quantization level.
1. I don't fully understand the fourth step you mentionned concerning the recording in 8 bit mode instead of 16 (use a property node to choose which 8 bits are most significant ). Where should the property node be located ? Actually, how could the significant bits be chosen in the VI ? By chance, would you have a VI using 8 bits mode to provide as an example?
2. Would it be possible to lower the quantization level to 4 bits or even 2 bits (in order to maximize the instantaneous bandwidth)?
3. Could the procedure be transposed to the USRP Rio ?
Thanks a lot for your answer,
Best regards,
Laura.
Hello Erik,
thank you very much for this nice experiment! I would like to reproduce your GPS record/replay setup for students of signals and systems theory course. For this, I would like to kindly ask you about your RF link budget. Can you please describe the particular parts that were used in the RX and TX paths? I mean their gain, NF, and bandwidth or catalog numbers, so I can check that by myself.
It seems to me the RX gain briefly suggested above may not be enough the make the noise-like input signal appearing as 1 Vpp swing over the ADC. On the other hand, you may have encountered practical difficulties with some strong interfering signals that required lowering the input gain to prevent some kind of nonlinear distortion. Anyway, I would really appreciate a short comment on this.
Also, please, can you tell me what the RF frontend module of NI USRP 2920 is? Is that something from Ettus portfolio? In particular, we have N210 with UBX 40 which seems to be roughly comparable with the USRP 2920 bundle. Or am I wrong?
Kind regards,
Tom
N210 and WBX-40 will act as the NI USRP-2920 with the right firmware upgrade. The firmware installs with the driver and you can follow the instructions in the getting started guide.
Hi Erik,
thank you very much for your comment.
The thing is I have UBX-40 (i.e. this https://www.ettus.com/product/details/UBX40 ) not WBX. Should it still work?
Kind regards,
Tom
Working for me, N210 with WBX (50-2200MHz) and VERT400 antenna
I am using USRP 2953R+PXI 8880+MXIx4 to do the record and playback system.
WLAN signal/Signal Bandwidth 80Mhz = IQ rate 100M, data size per second=100M*2*2B=400 MB/s (Is the calculation right?)
For the playback part, I tried for a long time, but I always got underflow error.
I make sure that the data in the queue is enough to supply USRP write, but still got error.
So, I tried to run the USRP example code “niUSRP EX Tx Continuous Async”.
Even the data already generated before the while loop, the underflow error still occurred.
I have no idea why the error always occurred.
What is the maximum BW rate can this example achieve? Is there any HW limitation? If I use 8260 SSD version, could I solve this issue?
I know USRP RIO can achieve 200MHz BW with PXI extender, Why “niUSRP EX Tx Continuous Async” example code cannot work?
Hello
i want to know what is difference between Data input to " configuration block of USRP "
and output wires called "coerced "
i note that it return same values
is they used or its just indicator
but if it indicator i input numbers why they shown
Chloevivian: I think transmitting always goes with a lower IQ rate, my USRP N210 has a theoretical bandwidth of 20M, but the max Rx IQ rate is 5M and Tx IQ RATE= 1M (sometimes even 100k)
The overflow errros are most of the time due to the computer limitations, if you're USRP has no FPGA on board (so if you run the code on your pc) then your PC and the connection are the bottleneck... try with lower settings or another computer?
M_Shaban: the "coerced" is indeed an indicator. It shows the actual transmitted/received frequency.
example: if you try to send a signal on frequency 5Ghz, but your daughterboard only supports up to 2Ghz, the transmitted signal will be 2Ghz, this will also be shown on the coerced indicator because it's the maximum value the hardware supports.
Hello,
When playing back a recorded signal, do the parameters have to be the same for IQ rate and Write Buffer? When I am playing back, I get a lot of packet loss errors and underflow errors. Does anyone have any insight as to why?
I am recording a GPS signal from the Spirent with the following set parameters:
carrier frequency: 1.57542g
IQ rate: 10Ms/s
gain: 0
Write Buffer: 200000
Thank you!
10 MS/s is likely too high for your computer. 5MS/s is sufficient for GPS.
Also be sure you have a more stable clock source for better lock.
From: Monkeydoo <web.community@ni.com>
Reply-To: "ni-169410736-9iy-2x-130w@community-mail.ni.com" <ni-169410736-9iy-2x-130w@community-mail.ni.com>
Date: Tuesday, August 16, 2016 at 5:01 PM
To: Erik Luther <erik.luther@ni.com>
Subject: Re: - RF Record and Playback with NI USRP
NI Community<https://decibel.ni.com/content/index.jspa>
RF Record and Playback with NI USRP
new comment by Monkeydoo<https://decibel.ni.com/content/people/Monkeydoo> View all comments on this document<https://decibel.ni.com/content/docs/DOC-22174#comment-50576
Thanks for your reply. I will try 5Ms/s. I am using a 10MHz ref signal. However, in the test lab that this is going to, they do not have a 10MHz reference. Can an internal reference be used on the USRP?
Usrp's tcxo isn't quite stable enough for a good lock. You can even use a function gen instrument with a 10 mhz sine wave.
Regards,
Erik
Sent from mobile device.
Just to clarify, I should record and playback using 5Ms/s? Or can I record
at 10Ms/s and playback at 5Ms/s?
Its like a tape recorder / record player. You can play back faster but it affects the frequency.
Unless you resample the signal you'll create time and frequency changes. You can test this with fm radio record and playback. They will sound like Alvin and the chipmunks.
For gps using 2 rates without digital resample won't work because it is digital modulation.
Regards,
Erik
Sent from mobile device.
Thank you. Well, I tried 5Ms/s and 50,000 , 100,000 and 200,000 IQ samples
and it still crashes while on the playback. I'm only recording 3 minutes.
Any ideas?
Hello Erik,
Does this vi support USB USRP devices such as the B200? I was issued a B200 but have access to several other USRP models.
I tried on 3 different PCs, the last two being i7 core processors with
different gigabit Ethernet cards and they all keep crashing with packet
loss or tx underflow errors. I tried lowering the IQ rate to 5MS/s from
10MS/s and decreased the write buffer in the playback to 40,000. Any ideas?
Thanks.
Hi
Is it possible to Play from the file and record to the file at the same time using Loop back mode (TX1 with SMA to RX1 with SMA cable).
I tried this, able to record with no error and not able to playback once recording is finished. Not getting any audio but waveform data getting same.
Could you please help us on this?
Thank you.
Hi Can someone help.. I am not able to see any Signal while recording GPS using USRP 2922.. I am using active antenna 20 dB gain+ Bias Tee + external LNA 13 dB gain.. still no preamble could be found.. Further, I am not able to understand how we can record data in I8 format
in place of I16.. niUSRP property node has no option of I8 as far as I tried. I tried using data typecasting from I16 to I8 but it subtracts 256 from all I16 samples only.. I do not know how that will be useful.. can any one help in getting records in I8 format also how to record GPS data using USRP 2922 or 2932.. how much external gain will be required..?

Hello everyone,
I have some doubts on I16 to I8 conversion in GPS record and replay .vi
1. How to change the MSRP Read and write to 8 bit.
2. How to change the queues used to store data to the 8-bit mode by changing the datatypes to I8
Hello Erik,
I want to test my GPS receiver and i have NI URSP.
Can you please share your recorded GPS bin file.
Thanks
Vikram kumar P
I am using LAB view 2020 64 bit with USRP Ni 2900
currently i am using another NI USRP Record and Playback - I16.llb 219 KB
I am using RX2 for recording through an active antenna mounted on roof with a feeded bias tee
it is making .bin file but not showing any GPS satellite
I m suing 1575.42M, IQ rate 4M and gain 20dB
Dear All,
I am new to NI USRP SDRs. I am facing issues. can any one help me. This FM demodulation is not working.